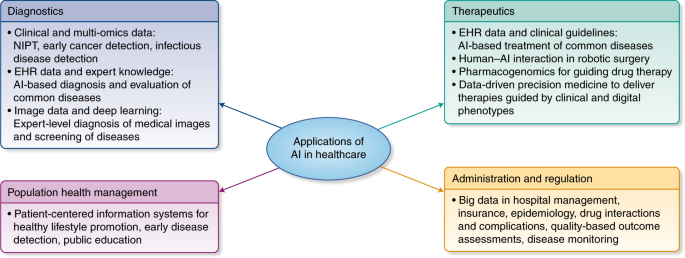
A Framework for the Design of Risk-Adjustment Models in Health Care Provider Payment Systems
A partir d'avui aquest blog es trasllada a Substack. Durant unes setmanes serà accessible simultàniament per blogger i per substack. Anoteu l'adreça: econsalut.substack.com
Article resumit amb IA.
Aquest article presenta un marc conceptual integral per al disseny de models d'ajust de risc (RA) en el context de models de pagament prospectiu a proveïdors d'assistència sanitària. L'objectiu és desenvolupar un marc que expliciti les opcions de disseny i les compensacions associades per tal de personalitzar el disseny de l'RA als sistemes de pagament a proveïdors, tenint en compte els objectius i les característiques del context d'interès.
Introducció (1-3): Durant les últimes dècades, els reguladors i els responsables polítics de la salut han fet esforços per millorar l'eficiència de la prestació d'assistència sanitària mitjançant la reforma dels sistemes de pagament a proveïdors. Específicament, l'eficiència s'ha perseguit mitjançant la introducció d'elements prospectius en els models de pagament, donant lloc a diversos Models de Pagament Alternatius (MPA) com els acords de qualitat alternatius i els pagaments agrupats. Aquests MPA tenen com a objectiu incentivar l'eficiència traslladant (part de) la responsabilitat financera dels pagadors als proveïdors. Una característica típica dels pagaments prospectius a proveïdors és que es basen en un "nivell de despesa normatiu" per a la prestació d'un conjunt predefinit de serveis a una determinada població de pacients. El nivell de despesa normatiu es refereix al nivell de despesa que "hauria de ser" depenent de la població de pacients d'un proveïdor, en lloc de la despesa observada. Un element clau en la determinació dels nivells de despesa normatius és la correcció de les diferències sistemàtiques en les necessitats d'assistència sanitària de les poblacions de pacients dels proveïdors, comunament coneguda com a ajust de risc (RA). L'RA és crucial per garantir un terreny de joc igualitari per als proveïdors i per evitar incentius per a comportaments no desitjats, com la selecció de riscos.
Nova Contribució (8-10): Tot i les contribucions conceptuals existents sobre el disseny de l'RA, actualment no hi ha un marc integral per adaptar el disseny de l'RA al pagament de proveïdors i a les característiques essencials del context. Aquest article desenvolupa aquest marc sintetitzant, ampliant i aplicant coneixements de la literatura existent. La metodologia va incloure una revisió de la literatura combinada amb consultes a experts en el camp de l'RA i els sistemes de pagament. La informació recopilada es va sintetitzar per desenvolupar el marc, del qual van sorgir tres criteris per al disseny de models d'RA i es van agrupar les opcions i les compensacions en dues dimensions principals: (a) la tria dels ajustadors de risc i (b) la tria de les ponderacions de pagament.
Definicions de Conceptes Clau (11-13): Els models de pagament prospectiu i els MPA traslladen la responsabilitat financera dels pagadors als proveïdors per tal d'incentivar el control de costos i l'eficiència. Qualsevol trasllat de responsabilitat financera requereix que el pagador determini el nivell de despesa normatiu, que reflecteix el nivell de despesa apropiat donades les necessitats d'assistència sanitària d'una població i els objectius dels MPA. El nivell de despesa normatiu no fa referència necessàriament al nivell de despesa absolut o òptim, sinó al nivell considerat apropiat donat el nivell/objectius d'eficiència perseguits pel MPA.
Fonts de Variació de la Despesa i el Paper de l'RA i la Mancomunació de Riscos (14-19): Quan s'estableixen nivells de despesa normatius, és important considerar tres fonts de variació de la despesa: (a) variació sistemàtica impulsada per factors fora del control dels proveïdors (variables C o "factors de compensació"), (b) variació sistemàtica impulsada per factors que els proveïdors poden influir (variables R o "factors de responsabilitat"), i (c) variació aleatòria. Per evitar que els proveïdors assumeixin riscos excessius que no poden influir, els MPA solen aplicar alguna forma de mancomunació de riscos. L'RA prospectiu s'utilitza per compensar la variació de la despesa deguda a les variables C. La naturalesa i el grau en què s'ha de compensar la variació de la despesa resultant de les variables C forma el punt de partida d'un model d'RA.
Tres Criteris per al Disseny de Models d'RA (19-26): L'objectiu general de l'RA en els MPA és compensar els proveïdors per la variació de la despesa deguda a les variables C, alhora que els manté responsables de la variació de la despesa deguda a les variables R. Això implica dos criteris clau: (a) compensació adequada per a les variables C i (b) cap compensació per a les variables R. Un tercer criteri important és la viabilitat.
- Criteri 1: Compensació Adequada per a les Variables C (20-26): Per evitar problemes de selecció, l'RA hauria de compensar adequadament les variables C que són rellevants a la llum de les possibles accions de selecció de riscos per part dels proveïdors (atraure/dissuadir pacients sans/no sans). També hauria de compensar les variables C que varien entre les poblacions de proveïdors per evitar la participació selectiva en el MPA.
- Criteri 2: Cap Compensació per a les Variables R (26-29): Per evitar ineficiències, l'RA no hauria de compensar les variables R. La compensació per la variació de la despesa de les variables R pot donar lloc a problemes d'eficiència, com la perpetuació de les ineficiències existents ("biaix d'status quo") i la creació d'incentius per a noves ineficiències (reducció dels incentius per al control de volum i preu, codificació ascendent).
- Criteri 3: Viabilitat (29-30): Un tercer criteri crucial és la viabilitat, que inclou la disponibilitat de dades i l'acceptació per part de totes les parts interessades (pacients, proveïdors, pagadors, reguladors).
Un Marc per al Disseny de Models d'RA (30-31): Aquest marc distingeix entre preguntes de disseny, opcions associades i consideracions i compensacions clau pel que fa a (a) la tria dels ajustadors de risc i (b) la tria de les ponderacions de pagament.
La Tria dels Ajustadors de Risc (31-47): Aquesta secció aborda tres preguntes principals de disseny:
- Quin tipus d'informació es basa els ajustadors de risc? (32-38): Les opcions inclouen informació demogràfica, socioeconòmica, subjectiva (de salut), diagnòstica, d'utilització, clínica, de despesa (retardada) i del costat de l'oferta. L'ús d'informació endògena (diagnòstics, utilització, despesa) és altament predictiu de la despesa de tipus C, però pot perpetuar ineficiències i introduir nous incentius perversos per al volum i el preu. L'ús d'informació exògena (demogràfica, socioeconòmica) no manté ni introdueix incentius perversos relacionats amb el volum o el preu, però el seu poder predictiu és generalment baix.
- A quin període de temps (període base) pertany la informació? (38-45): Es pot distingir entre ajustadors concurrrents i prospectius. Els efectes d'incentiu relatius d'aquestes opcions no estan clars a priori.
- Com dissenyar els ajustadors de risc? (46-47): Això inclou l'especificació de l'escala de mesura, l'operacionalització dels ajustadors (considerant condicions, jerarquies, restriccions) i les interaccions entre ajustadors.
La Tria de les Ponderacions de Pagament (48-60): Per trobar ponderacions de pagament apropiades, els responsables de la presa de decisions s'enfronten a tres decisions principals de disseny:
- Quina mostra d'estimació? (49-52): Es requereix una mostra d'estimació representativa de la població d'interès i dels nivells de despesa normatius. En la pràctica, sovint s'utilitzen dades històriques i poblacions de pacients similars.
- Quines intervencions de dades? (52-58): Quan la mostra d'estimació no és representativa, s'han de considerar intervencions de dades sobre la població de pacients i/o les dades de despesa per millorar la coincidència amb la població d'interès i el nivell de despesa normatiu. Això pot incloure correccions per biaixos i inequitats.
- Com derivar les ponderacions de pagament? (59-60): Això implica decidir quins ajustadors de risc incloure (considerant el biaix de la variable omesa) i quin criteri d'optimització utilitzar per estimar aquestes ponderacions. Les opcions van des de criteris d'optimització estàndard (OLS, GLM) fins a criteris personalitzats (regressió restringida, aprenentatge automàtic).
La Interconnexió Entre les Opcions de Disseny per als Ajustadors de Risc i les Ponderacions de Pagament (61-62): Les decisions de disseny dins i entre aquests dos temes estan altament interrelacionades. Per exemple, la tria de la informació en què es basen els ajustadors de risc afectarà la seva especificació i operacionalització. De la mateixa manera, les decisions sobre com es deriven les ponderacions de pagament depenen tant de la tria dels ajustadors de risc com de la tria de la mostra d'estimació (modificada).
Discussió (63-68): No hi ha un enfocament únic per al disseny de models d'RA, i el disseny adequat pot variar segons la configuració i les evolucions al llarg del temps. És crucial la decisió normativa sobre quines variables es consideren C i quines R. L'abast de la preocupació pels possibles incentius de selecció i control de costos pot variar segons el context. Les consideracions de viabilitat, com la disponibilitat de dades i l'acceptació de les parts interessades, també són importants.
Consideracions Més Amplies per al Disseny de l'RA en el Finançament de l'Assistència Sanitària (69-70): Tot i que aquest article se centra en el pagament a proveïdors, el marc proposat també podria beneficiar altres reformes de finançament, com les iniciatives de participació del consumidor, tot i que es necessita més recerca.
Conclusió (71): El disseny de models d'RA per a sistemes de pagament prospectiu a proveïdors és un exercici complex que requereix una consideració explícita de moltes preguntes, opcions i compensacions difícils. El procés de disseny ha de guiar-se per tres criteris clau: compensació adequada de les variables C, cap compensació de les variables R i viabilitat. Les diverses preguntes i opcions de disseny es poden classificar en la tria dels ajustadors de risc i la tria de les ponderacions de pagament. Es necessita més recerca per donar suport a les decisions normatives sobre les variables C i R, així com per desenvolupar mètriques d'avaluació integrals per a la valoració dels efectes dels incentius.
Referències
A continuació es mostren les referències citades en les fonts:
- Adams Dudley, R., Medlin, C. A., Hammann, L. B., Cisternas, M. G., Brand, R., Rennie, D. J., & Luft, H. S. (2003). The best of both worlds? Potential of hybrid prospective/concurrent risk adjustment. Medical Care, 41(1), 56–69.
- American Medical Association. (2019). Improving Risk adjustment in alternative payment models.
- Andersen, R., & Newman, J. F. (2005). Societal and individual determinants of medical care utilization in the United States. The Milbank Memorial Fund Quarterly. Health and Society, 83(4), 1468-0009.2005.00428.x.
- Anderson, G. F., & Weller, W. E. (1999). Methods of reducing the financial risk of physicians under capitation. Archives of Family Medicine, 8(2), 149–155.
- Anthun, K. S. (2021). Predicting diagnostic coding in hospitals: Individual level effects of price incentives. International Journal of Health Economics and Management, 22(2), 129–146.
- Arrow, K. J. (2004). Uncertainty and the welfare economics of medical care. Bulletin of the World Health Organization, 82(2), 141–149.
- Ash, A. S., & Ellis, R. P. (2012). Risk-adjusted payment and per-formance assessment for primary care. Medical Care, 50(8), 643–653.
- Ash, A. S., Mick, E. O., Ellis, R. P., Kiefe, C. I., Allison, J. J., & Clark, M. A. (2017). Social determinants of health in managed care payment formulas. JAMA Internal Medicine, 177(10), 1424–1430.
- Bäuml, M. (2021). How do hospitals respond to cross price incen-tives inherent in diagnosis-related groups systems? The impor-tance of substitution in the market for sepsis conditions. Health Economics, 30(4), 711–728.
- Bergquist, S. L., Layton, T. J., McGuire, T. G., & Rose, S., & National Bureau of Economic Research. (2018). Intervening on the data to improve the performance of health plan pay-ment methods (Ser. NBER working paper series, no. w24491). National Bureau of Economic Research.
- Brown, J., Duggan, M., Kuziemko, I., & Woolston, W. (2014). How does risk selection respond to risk adjustment? New evi-dence from the Medicare Advantage Program. The American Economic Review, 104(10), 3335–3364.
- Buchner, F., Wasem, J., & Schillo, S. (2017). Regression trees iden-tify relevant interactions: Can this improve the predictive per-formance of risk adjustment? Health Economics, 26(1), 74–85.
- Cattel, D., Eijkenaar, F., & Schut, F. T. (2020). Value-based pro-vider payment: Towards a theoretically preferred design. Health Economics, Policy and Law, 15(1), 94–112.
- Chang, H.-Y., Lee, W.-C., & Weiner, J. P. (2010). Comparison of alternative risk adjustment measures for predictive modeling: High risk patient case finding using Taiwan’s national health insurance claims. BMC Health Services Research, 10, 343– 343. https://doi.org/10.1186/1472-6963-10-343.
- Chernew, M. E., Mechanic, R. E., Landon, B. E., & Safran, D. G. (2011). Private-payer innovation in Massachusetts: The “alter-native quality contract.” Health Affairs, 30(1), 51–61.
- Chien, A. T., Newhouse, J. P., Iezzoni, L. I., Petty, C. R., Normand, S.-L. T., & Schuster, M. A. (2017). Socioeconomic background and commercial health plan spending. Pediatrics, 140(5).
- Constantinou, P., Tuppin, P., Gastaldi-Ménager, C., & Pelletier-Fleury, N. (2022). Defining a risk-adjustment formula for the introduction of population-based payments for primary care in France. Health Policy, 126(9), 915–924.
- Dafny, L. S. (2005). How do hospitals respond to price changes? The American Economic Review, 95(5), 1525–1547.
- Douven, R., McGuire, T. G., & McWilliams, J. M. (2015). Avoiding unintended incentives in ACO payment models. Health Affairs, 34(1), 143–149.
- Douven, R., Remmerswaal, M., & Mosca, I. (2015). Unintended effects of reimbursement schedules in mental health care. Journal of Health Economics, 42, 139–150.
- Dowd, B. E., Huang, T-y, & McDonald, T. (2021). Tiered cost-sharing for primary care gatekeeper clinics. American
- Duan, N., Manning, W. G., Morris, C. N., & Newhouse, J. P. (1983). A comparison of alternative models for the demand for medical care. Journal of Business & Economic Statistics, 1(2), 115–126.
- Durfey, S. N. M., Kind, A. J. H., Gutman, R., Monteiro, K., Buckingham, W. R., DuGoff, E. H., & Trivedi, A. N. (2018). Impact of risk adjustment for socioeconomic status on Medicare advantage plan quality rankings. Health Affairs, 37(7), 1065–1072.
- Eijkenaar, F., van Vliet, R. C. J. A., & van Kleef, R. C. (2018). Diagnosis-based cost groups in the Dutch risk-equalization model: Effects of clustering diagnoses and of allowing patients to be classified into multiple risk-classes. Medical Care, 56(1), 91–96.
- Einav, L., Finkelstein, A., Ji, Y., & Mahoney, N. (2022). Voluntary regulation: Evidence from Medicare payment reform. The Quarterly Journal of Economics, 137(1), 565–618.
- Ellis, R. P. (1998). Creaming, skimping and dumping: Provider competition on the intensive and extensive margins. Journal of Health Economics, 17(5), 537–555.
- Ellis, R. P., Martins, B., & Rose, S. (2018). Risk adjustment for health plan payment. In T. G. McGuire & R. C. van Kleef (Eds.), Risk adjustment, risk sharing and premium regu-lation in health insurance markets: Theory and practice (pp. 55–104). Elsevier.
- Epstein, A. M., & Cumella, E. J. (1988). Capitation payment: Using predictors of medical utilization to adjust rates. Health Care Financing Review, 10(1), 51–69.
- García-Goñi, M., Ibern, P., & Inoriza, J. M. (2009). Hybrid risk adjustment for pharmaceutical benefits. The European Journal of Health Economics, 10(3), 299–308.
- Geruso, M., & Layton, T. (2020). Upcoding: Evidence from Medicare on squishy risk adjustment. Journal of Political Economy, 128(3), 984–1026.
- Geruso, M., & McGuire, T. G. (2016). Tradeoffs in the design of health plan payment systems: Fit, power and balance. Journal of Health Economics, 47, 1–19.
- Gilmer, T., Kronick, R., Fishman, P., & Ganiats, T. G. (2001). The Medicaid Rx model: Pharmacy-based risk adjustment for pub-lic programs. Medical Care, 39(11), 1188–1202.
- Glazer, J., & McGuire, T. G. (2002). Setting health plan premiums to ensure efficient quality in health care: Minimum variance optimal risk adjustment. Journal of Public Economics, 84(2), 153–173.
- Gravelle, H., Sutton, M., Morris, S., Windmeijer, F., Leyland, A., Dibben, C., & Muirhead, M. (2003). Modelling supply and demand influences on the use of health care: Implications for deriving a needs-based capitation formula. Health Economics, 12(12), 985–1004.
- Hayen, A. P., van den Berg, M. J., Meijboom, B. R., Struijs, J. N., & Westert, G. P. (2015). Incorporating shared savings pro-grams into primary care: From theory to practice. BMC Health Services Research, 15, Article 580.
- Heckman, J. J., & Honoré, B. E. (1990). The empirical content of the Roy model. Econometrica, 58(5), 1121–1149.
- Hildebrandt, H., Hermann, C., Knittel, R., Richter-Reichhelm, M., Siegel, A., & Witzenrath, W. S. (2010). Gesundes Kinzigtal Integrated Care: Improving population health by a shared health
- Hollenbeak, C. S. (2005). Functional form and risk adjustment of hospital costs: Bayesian analysis of a Box-Cox random coef-ficients model. Statistics in Medicine, 24(19), 3005–3018. https://doi.org/10.1002/sim.2172.
- Horn, S. D., Horn, R. A., & Sharkey, P. D. (1984). The severity of illness index as a severity adjustment to diagnosis-related groups. Health Care Financing Review, 1984, 33–45.
- Hughes, J. S., Averill, R. F., Eisenhandler, J., Goldfield, N. I., Muldoon, J., Neff, J. M., & Gay, J. C. (2004). Clinical risk groups (CRGs): A classification system for risk-adjusted capitation- based payment and health care management. Medical Care, 42(1), 81–90.
- Humbyrd, C. J., Hutzler, L., & DeCamp, M. (2019). The ethics of success in bundled payments: Respect, beneficence, and social justice concerns. American Journal of Medical Quality, 34(2), 202–204.
- Jha, A. (2014) The Health Care Blog. https://thehealthcareblog. com/blog/2014/09/29/changing-my-mind-on-ses-adjustment/
- Iezzoni, L. I. (2012). Risk adjustment for measuring health care outcomes (3rd ed.). Health Administration Press.
- Iommi, M., Bergquist, S., Fiorentini, G., & Paolucci, F. (2022). Comparing risk adjustment estimation methods under data availability constraints. Health Economics, 31(7), 1368–1380.
- Isaksson, D., Blomqvist, P., Pingel, R., & Winblad, U. (2018). Risk selection in primary care: A cross-sectional fixed effect analysis of Swedish individual data. BMJ Open, 8(10), Article e020402.
- Jones, A. (2010). Models for health care [Working papers]. HEDG, c/o Department of Economics, University of York, Health, Econometrics and Data Group (HEDG).
- Kapur, K., Tseng, C. W., Rastegar, A., Carter, G. M., & Keeler, E. (2003). Medicare calibration of the clinically detailed risk information system for cost. Health Care Financing Review, 25(1), 37–54.
- Lamers, L. M. (1998). Risk-adjusted capitation payments: Developing a diagnostic cost groups classification for the Dutch situation. Health Policy, 45(1), 15–32.
- Lamers, L. M. (1999). Pharmacy costs groups. Medical Care, 37(8), 824–830.
- Lamers, L. M., & van Vliet, R. C. J. A. (2003). Health-based risk adjustment; Improving the pharmacy-based cost group model to reduce gaming possibilities. European Journal of Health Economics, 4(2), 107–114.
- Layton, T. J., McGuire, T. G., & van Kleef, R. C. (2018). Deriving risk adjustment payment weights to maximize efficiency of health insurance markets. Journal of Health Economics, 61, 93–110.
- Lemke, K. W., Pham, K., Ravert, D. M., & Weiner, J. P. (2020). A revised classification algorithm for assessing emergency department visit severity of populations. American Journal of Managed Care, 26(3), 119–125. https://doi.org/10.37765/ ajmc.2020.42636.
- Levy, S., Bagley, N., & Rajkumar, R. (2018). Reform at risk— mandating participation in alternative payment plans. The New England Journal of Medicine, 378(18), 1663–1665.
- Liao, J. M., Pauly, M. V., & Navathe, A. S. (2020). When should Medicare mandate participation in alternative payment mod-els? Health Affairs, 39(2), 305–309.
- Martinussen, P. E., & Hagen, T. P. (2009). Reimbursement sys-tems, organisational forms and patient selection: Evidence from day surgery in Norway. Health Economics, Policy, and Law, 4(Pt. 2), 139–158.
- McGuire, T. G. (2000). Physician agency. In A. J. Culter & J. P. Newhouse (Eds.), Handbook of health economics (1st ed., Vol. Volume 1a, Handbooks in economics, p. 17). Elsevier.
- McGuire, T. G. (2011) Physician agency and payment for primary medical care. In S. Glied & P. C. Smith (Eds), Chapter 9: The Oxford handbook of health economics (online ed., pp. 462– 528). Oxford Academic.
- McWilliams, J. M., Hatfield, L. A., Landon, B. E., Hamed, P., & Chernew, M. E. (2018). Medicare Spending after 3 years of the Medicare shared savings program. The New England Journal of Medicine, 379(12), 1139–1149.
- McWilliams, J. M., Weinreb, G., Ding, L., Ndumele, C. D., & Wallace, J. (2023). Risk adjustment and promoting health equity in population-based payment: Concepts and evidence: Study examines accuracy of risk adjustment and payments in promoting health equity. Health Affairs, 42(1), 105–114.
- Newhouse, J. P. (1994). Patients at risk: Health reform and risk adjustment. Health Affairs, 13(1), 132–146.
- Newhouse, J. P., Price, M., McWilliams, J. M., Hsu, J., & McGuire, T. G. (2015). How much favorable selection is left in Medicare advantage? American Journal of Health Economics, 1(1), 1– 26. https://doi.org/10.1162/ajhe_a_00001.
- Politzer, E. (2024). Utilization thresholds in risk adjustment sys-tems. American Journal of Health Economics, 10(3), 470–503.
- Pope, G. C., Kautter, J., Ellis, R. P., Ash, A. S., Ayanian, J. Z., Lezzoni, L. I., Ingber, M. J., Levy, J. M., & Robst, J. (2004). Risk adjustment of Medicare capitation payments using the CMS-HCC model. Health Care Financing Review, 25(4), 119–141.
- Pope, G. C., Kautter, J., Ingber, M. J., Freeman, S., Sekar, R., & Newhart, C. (2011). Evaluation of the CMS-HCC Risk Adjustment Model [Prepared by RTI]. Centers for Medicare & Medicaid Services, Medicare Plan Payment Group, Division of Risk Adjustment and Payment Policy.
- Porter, M. E., & Kaplan, M. S. (2016). How to pay for health care? Harvard Business Review, 94(7/8), 88–102.
- Robinson, J. C., Whaley, C., Brown, T. T., & Dhruva, S. S. (2020). Physician and patient adjustment to reference pricing for drugs. JAMA Network Open, 3(2), Article e1920544.
- Rose, S. (2016). A machine learning framework for plan payment risk adjustment. Health Services Research, 51(6), 2358–2237.
- Rose, S., Zaslavsky, A. M., & McWilliams, J. M. (2016). Variation in accountable care organization spending and sensitivity to risk adjustment: Implications for benchmarking. Health Affairs, 35(3), 440–448.
- Schokkaert, E., & Van de Voorde, C. (2004). Risk selection and the specification of the conventional risk adjustment formula. Journal of Health Economics, 23, 1237–1259.
- Schokkaert, E., & Van de Voorde, C. (2006). Incentives for risk selection and omitted variables in the risk adjustment formula. Annales d’economie et de Statistique, 83-84, 327–352.
- Struijs, J. N., de Vries, E. F., Baan, C. A., van Gils, P. F., & Rosenthal, M. B. (2020). Bundled-payment models around the world: How they work and what their impact has been [Issue brief]. The Commonwealth Fund.
- van Barneveld, E. M., Lamers, L. M., van Vliet René, C. J. A., & van de Ven, W. P. M. M. (2001). Risk sharing as a supplement to imperfect capitation: A tradeoff between selection and effi-ciency. Journal of Health Economics, 20(2), 147–168.
- van de Ven, W. P. M. M., & Ellis, R. P. (2000). Risk adjustment in competitive health plan markets. In A. J. Collyer & J. P. Newhouse (Eds.), Handbook of health economics (pp. 1003– 1092). Elsevier Science.
- van de Ven, W. P. M. M., & van Vliet, R. C. J. A. (1992). How can we prevent cream skimming in a competitive health insur-ance market? The great challenge for the ‘90’s. In P. Zweifel & H. E. French (Eds.), Health economics worldwide (pp. 23–46). Kluwer Academic Publishers.
- van Kleef, R. C., Eijkenaar, F., van Vliet René, C. J. A., & Nielen, M. M. J. (2020). Exploiting incomplete information in risk adjustment using constrained regression. American Journal of Health Economics, 6(4), 477–497.
- van Kleef, R. C., McGuire, T. G., van Vliet René, C. J. A., & van de Ven, W. P. P. M. (2017). Improving risk equalization with constrained regression. The European Journal of Health Economics: Health Economics in Prevention and Care, 18(9), 1137–1156.
- van Kleef, R. C., & Reuser, M. (2021). How the covid-19 pan-demic can distort risk adjustment of health plan payment. The European Journal of Health Economics, 22(7), 1005–1016.
- van Kleef, R. C., & van Vliet René, C. J. A. (2012). Improving risk equalization using multiple-year high cost as a health indicator. Medical Care, 50(2), 140–144.
- Veen, S. H. C. M., Kleef, R. C., Ven, W. P. M. M., & Vliet, R. C. J. A. (2018). Exploring the predictive power of interaction terms in a sophisticated risk equalization model using regres-sion trees. Health Economics, 27(2), 12.
- Vermaas, A. (2006). Agency, managed care and financial-risk sharing in general medical practice [Dissertation]. Erasmus Universiteit.
- Werbeck, A., Wübker, A., & Ziebarth, N. R. (2021). Cream skim-ming by health care providers and inequality in health care access: Evidence from a randomized field experiment. Journal of Economic Behavior and Organization, 188, 1325–1350.
- Withagen-Koster, A. A., van Kleef, R. C., & Eijkenaar, F. (2020). Incorporating self-reported health measures in risk equaliza-tion through constrained regression. The European Journal of Health Economics, 21(4), 513–528.



.jpg)