CRISPR: Genome Editing and Engineering And Related Issues
15 d’octubre 2022
14 d’octubre 2022
Pandemethics (3)
The Ethics of Pandemics. An Introduction
Table of Contents
1. Introduction: Why Ethics of Pandemics?
2. The General Principle of Pandemic Response
3. Rationing of Scarce Health Care Resources
4. Pandemics in an Unequal World
5. Restricting Freedom
6. Inducing Voluntary Behavioral Change
7. Moral Mathematics under Uncertainty.
13 d’octubre 2022
The architects of choice
How “nudges” by government can empower citizens without manipulating their preferences or exploiting their biases.
We're all familiar with the idea of “nudging”—using behavioral mechanisms to encourage people to make certain choices—popularized by Richard Thaler and Cass Sunstein in their bestselling 2008 book Nudge. This approach, also known as “libertarian paternalism,” goes beyond typical programs that simply provide information and incentives; nudges can range from automatic enrollment in a pension plan to flu-shot scheduling. In Nudging, Riccardo Viale explores the evolution of nudging and proposes new approaches that would empower citizens without manipulating them paternalistically. He shows that we can use the tools of the behavioral sciences without abandoning the principle of conscious decision-making.
12 d’octubre 2022
Pharma, big pharma (16)
The Truth About the Drug Companies. HOW THEY DECEIVE US AND WHAT TO DO ABOUT IT
From the former editor-in-chief of the New England Journal of Medicine and now a member of Harvard Medical School’s Department of Global Health and Social Medicine, Marcia Angell
11 d’octubre 2022
The decline of pharma R&D productivity (2)
Science needs to move beyond luck if it is to design better drugs for the brain
The Economist:
Between 2011 and 2020 the likelihood of a drug in psychiatry being approved by the Food and Drug Administration was 7.3%. In neurology it was 5.9%. (The industry average is 7.9%.) As well as being less likely to succeed in trials (see chart), neurology drugs also take much longer, on average, to develop, further decreasing their appeal
According to the Global Burden of Disease project 12 mental-health disorders affect about 970m people. Their prevalence has increased by 48% since 1990 as the population has grown. With more than one in ten people on the planet affected, it is a global problem, although what data are available suggest it is more marked in Western countries (see map).
10 d’octubre 2022
Paying for rare diseases drugs
The next generation of rare disease drug policy: ensuring both innovation and affordability
The study finds that while 5% of drugs with an orphan indication cost more than $500,000 per year in US, these drugs make up only 0.08% of all patients treated by drugs with an orphan indication. The majority (52%) of patients who receive treatment an orphan drug get the treatment for <$50,000.
09 d’octubre 2022
Pharmaceutical contracts and prices
Introduction to Market Access for Pharmaceuticals
Contents:
Chapter 1: Health as a Good
Chapter 2: Decision-Making in Public Health
Chapter 3: Definition and Concepts
Chapter 4: HTA Decision Analysis framework
Chapter 5: Early HTA Advice
Chapter 6: Overview of Market Access Agreements
Chapter 7: External Reference Pricing
Chapter 8: Gap between Payers and Regulators
Chapter 9: Early Access Programs
Chapter 10: Market Access of Orphan Drugs
08 d’octubre 2022
Building a think tank
Build a think tank. A guide for policy entrepreneurs
Using this guide 8The reasons for creating this companion guide 8
Using this companion guide 8
Introduction 11
The importance of supporting new think tank development 11
Unravelling the definition of think tanks 13
What’s in the name? 13
Defining think tanks 14
Think tank functions 15
Summing up 16
The Why? questions 18
Why do you want to set up a think tank? 18
Why do think tanks aim to influence policy? 23
Are you sure you want to establish a think tank? 25
The What? questions 26
What will your think tank do? 26
What is the context? 27
What do you want to achieve by setting up a think tank? 33
What issues will the think tank focus on? 37
What will the think tank want to influence? 38
What will its business model be? 40
The Who? questions 46
Who will govern it? (And how?) 46
Who will lead the think tank? 51
Who will engage with it? 56
Who will work for it? 60
Who will fund it? 65
Who will support it? 71
The How? questions 72
How will it carry out research? 72
How will it be managed? 79
How will it communicate? 82
How will it monitor its progress? 88
How will you ensure its credibility? 92
How will it adapt to change? 95
How will it engage with evolving technology? 99
The ‘When’? questions 101
When to start? 101
When to let go? 106
Checklist: Establishing a think tank 108
References and resources 110
On the definition and functions of think tanks 110
Boards and governance 111
Creating and managing think tanks 111
Policy impact 111
Communications 112
Funding and financial management 112
Context 113
Evidence-informed policy 113
List of Boxes, Figures and Tables 114
07 d’octubre 2022
Market access for expensive therapies (or how to overcome prices) (2)
Gene and Cell Therapies-Market Access and Funding
Contents:
Chapter 1 Introduction to cell and gene therapies concepts and definitions in US and EU
Chapter 2 cell and gene therapies: genuine products and potential for dramatic value
Chapter 3 cell and gene therapies: Regulatory aspects in US and EU
Chapter 4 the need for new HTA reference case for cell and gene therapies
Chapter 5 How to mitigate cell and gene therapies uncertainties and HTA risk adverse attitude?
Chapter 6 Cell and gene therapies funding: challenges and solutions for patients’ access
Chapter 7 Conclusion
06 d’octubre 2022
Market access for expensive therapies (or how to overcome prices)
Managed Entry Agreements and Funding for Expensive Therapies
Key reference to understand what's going on in this topic. Recommended.
Contents:
1. Introduction to Managed Entry Agreements
2. Definition and Classification of MEAs
3. From Coverage with Evidence Development to Individual Performance-Based Agreements in Italy
4. Coverage with Evidence Development for Multiple Sclerosis Drugs in the UK: A "Costly Failure"?
5. Country Comparison of the Implementation of Managed Entry Agreements
6. Novel Funding Models for Expensive Therapies
7. Managed Entry Agreement for Cell and Gene Therapies
02 d’octubre 2022
The decline of pharma R&D productivity (how much are we willing to pay for failure (95%) or success (5%)?)
Global Trends in R&D . Overview through 2021
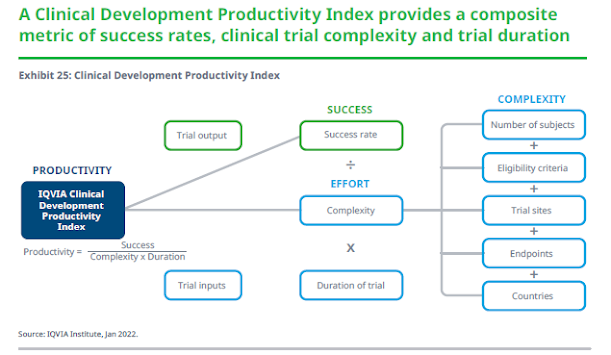
The productivity of the clinical development process can be considered as a measure of trial outputs (drugs, innovation, trial success, etc.), compared to a measure of trial inputs or resources dedicated to obtaining those outputs (e.g., aspects of trial complexity, duration, monetary investments, etc.). Such measures of success, complexity and trial duration were selected for inclusion in the productivity index as described above. Increases in success will increase productivity overall as will decreases in complexity or duration. Conversely, decreases in success will drive down the productivity index, as do increases in complexity and duration.
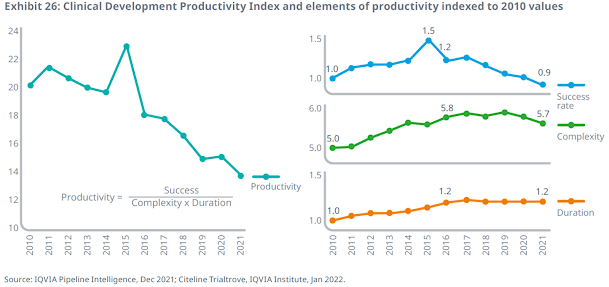
Clinical development productivity — a composite metric of success rates, clinical trial complexity and trial duration — declined in 2021, continuing an overarching 10-year trend. Despite a second year of decreasing trial complexity, the ongoing decline in success rates has resulted in reduced average trial productivity. Trial success rates fell to their lowest in more than 10 years to an average 5% likelihood of progressing successfully through all phases.
01 d’octubre 2022
Health systems design
Building a High- Value Health System
Countries and institutions worldwide face the challenge of planning and paying for health care that effectively meets the needs of citizens and employees. While there are many criticisms of existing healthcare models, current literature offers little guidance for individuals who want to carry out the work of redesigning and improving their health system.
29 de setembre 2022
22 de setembre 2022
Pharmaceutical innovation and value extraction
Pharmaceutical innovation sourcing
Figure below shows that 23% of new medicines came from public bodies and private-private collaboration and they didn't apply for any marketing authorisation.