Global Trends in R&D . Overview through 2021
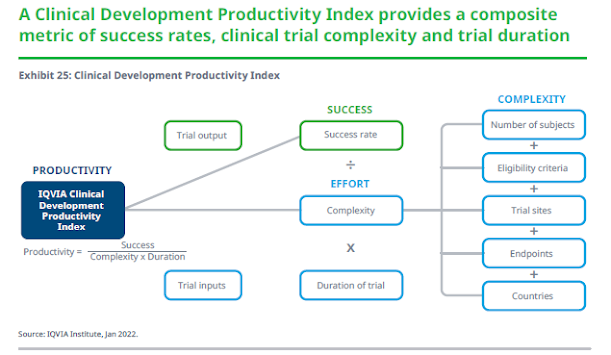
The productivity of the clinical development process can be considered as a measure of trial outputs (drugs, innovation, trial success, etc.), compared to a measure of trial inputs or resources dedicated to obtaining those outputs (e.g., aspects of trial complexity, duration, monetary investments, etc.). Such measures of success, complexity and trial duration were selected for inclusion in the productivity index as described above. Increases in success will increase productivity overall as will decreases in complexity or duration. Conversely, decreases in success will drive down the productivity index, as do increases in complexity and duration.
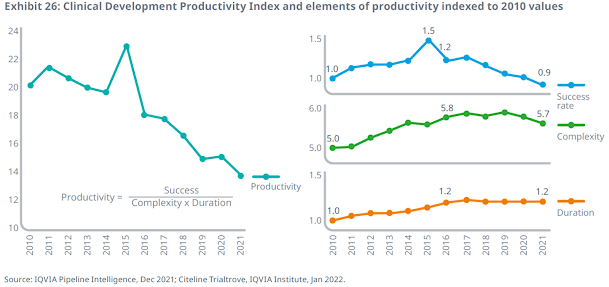
Clinical development productivity — a composite metric of success rates, clinical trial complexity and trial duration — declined in 2021, continuing an overarching 10-year trend. Despite a second year of decreasing trial complexity, the ongoing decline in success rates has resulted in reduced average trial productivity. Trial success rates fell to their lowest in more than 10 years to an average 5% likelihood of progressing successfully through all phases.