Some months ago in the best books of FT 2019, I listed this one. and the summary is:
The Pandemic Century exposes the limits of science against nature, and how these crises are shaped by humans as much as microbes.This is exactly what is happening right now!!! Contagion is shaped also by politicians.
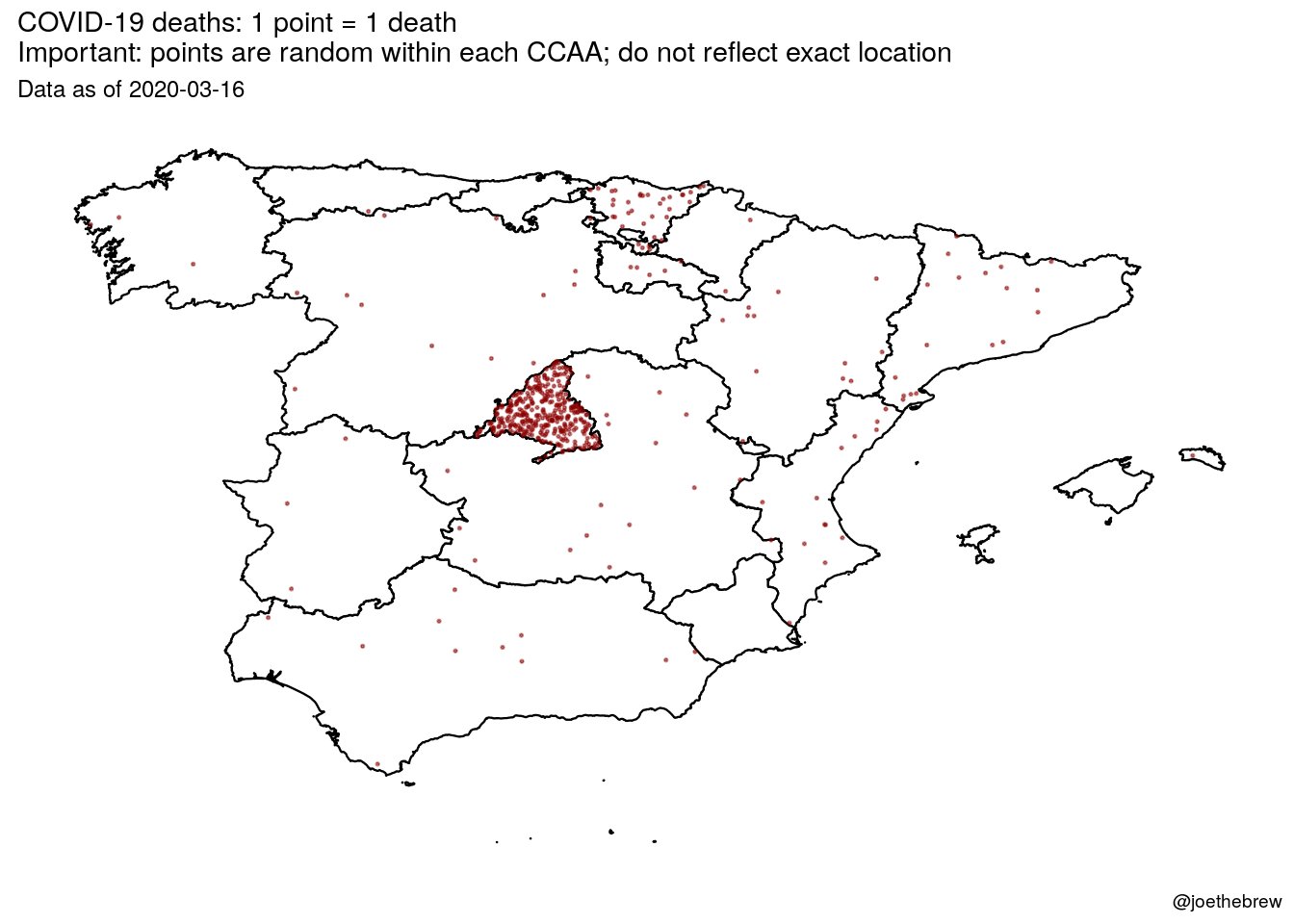
Just have a look at what's going on in Madrid. This is horrendous! And politicians don't want to close Madrid! And they have allowed to spread contagion outside. Somebody should say it louder. International health rules should prevail over spanish rulers.
Beyond politicians, there are experts.
Battered by their repeated failure to predict deadly outbreaks of infectious disease, even the experts have come to recognize the limits of medical prognostication. This is not only because microbes are highly mutable—that has been known since Pasteur’s time—but because we are continually lending them a helping a hand. Time and again, we assist microbes to occupy new ecological niches and spread to new places in ways that usually only become apparent after the event. And to judge by the recent run of pandemics and epidemics the process seems to be speeding up.
Reviewing the last hundred years of epidemic outbreaks, the only thing that is certain is that there will be new plagues and new pandemics. It is not a question of if, we are told, but when. Pestilences may be unpredictable but we should expect them to recur. However, what Camus could not have foreseen is that the attempt to anticipate disaster also creates new distortions and introduces new uncertainties. Twice this pandemic century, in 1976 and again in 2003, scientists thought the world was on the brink of a new influenza pandemic, only to realize that the outbreaks were false alarms and that the real danger lay elsewhere. Then in 2009 the WHO declared that the Mexican swine flu, a ressortment of two well-known H1N1 swine-lineage viruses that had circulated separately for over a decade, met the criterion of a pandemic virus, triggering the activation of global pandemic preparedness plans. On paper, this was the first pandemic of the twenty-first century and the first influenza pandemic in forty-one years. The fact that the swine flu was an H1N1, just like the Spanish flu, raised the prospect that this might be the Big One and that governments should expect a wave of illness and deaths similar to that in 1918–1919. But though the WHO’s declaration sparked widespread panic, the anticipated viral Armageddon never materialized. Instead, when it was realized that the Mexican swine flu was no more severe than a seasonal strain of flu, the WHO was accused of “faking” the pandemic for the benefit of vaccine manufacturers and other special interests. The result is what Susan Sontag calls “a permanent modern scenario: apocalypse looms . . . and it doesn’t occur.” As we look to the next one hundred years of infectious disease outbreaks, let us hope that is one prognostication that turns out to be true.